MEDICAL CARE IS OUR PROFESSION
We are a manufacturer of surgical disposable products for the area of infection control in
Operation Theater to industrial protective clothes which provide protecting health care to the end users.
We are a manufacturer of surgical disposable products for the area of infection control in
Operation Theater to industrial protective clothes which provide protecting health care to the end users.
From humble beginning 30 years ago to industry leader in Innovative disposable surgical products.
Stringent and International Standard like ISO 13485:2016 governs our productions.
Ensure consistent quality product.
hopes® a brand grown from consistent quality product from innovative technology and “customer focus”.

MEDIPRO (Malaysia) Sdn. Bhd. is a subsidiary of Japan Medical Products, Japan, was founded in year 1988 and located at Northport of Port Klang, Malaysia. Manufacturing of disposable medical device products which include Surgical Drape, Surgical Pack, Surgical Gown, Surgical Supporting Product, Surgical Cap and Mask for single use. MEDIPRO has enjoyed significant growth in its product range and customer base, with continual expansion both locally and internationally.
Being over 30 years in industry, MEDIPRO is recognized globally as a market leader.
We have our own distributor under our own brand - hopes®. We, the team, have 50-years experience in Japan and 30-years experience in Malaysian medical site. Our distributors are all across ASEAN countries such as Thailand, Myanmar, Singapore, Vietnam and Indonesia. MEDIPRO has a unique specialty that has been build up by the experiences with a huge number of the edged surgical-operations in Japan market, such as endoscopy, and we can give a good suitable solution to you, ASEAN hospitals. Now, many new users, ASEAN hospitals start to choose " hopes®", our protective products.
Quality assured by the means of very well planned and carefully put in place throughout our production line where it acts as gates.
The gates / checks are in the forms of simple visual checks to high tech equipment like latest sensors including incorporating Radio Frequency Identification (RFID) and scanners (QR codes).
This includes continuous and planned training of manpower development and education to produce more productivity and quality awareness.
Incorporating 5S routine to monitor and maintain the working environment for a healthy and productive workplace.
Our quality awareness and knowledge starts at the heart of company, our employees. The commitment to quality begins the day they join our family.
Our continuous commitment to achieve and deliver products of premium quality through accreditation from international recognised accredited bodies and conformity to the latest requirements for our products.
“Do it right the first time”and also incorporating Plan-Do-Check-Act to produce consistent premium products and “No delays”
To fulfil all our customers’ product requirements and committed to maintain the highest quality of product, material and delivery.

Medical Devices - Quality Management System (QMS). Requirements for regulatory purposes

Registered Medical Device Manufacturer with Medical Device Authority of Malaysia

Registration certificate for foreign medical device manufacturer with Ministry of Health, Japan

We aim to be an excellent manufacturing company through continuous improvement and change in the area of 5M + 1M (profitability) as our constant guide and foundation.

All regulatory affair dealing
Delegate customer complain & customer support
Develop and monitor sales plan and activities
Company financial accounting
LAN communication system
Human resource management
Corporate legal/ facilities
Company logistic/ warehouse/ packaging activities
Sales dealing/ services
Production overall activity
Manage production quality, rejection and loss
Material purchase activity
Product specification
Company business expansion
Quality and Validation
Quality Assurance
Validation
Lab, technical file & sterilization validation
Manage breakdown maintenance & preventive maintenance
Plan, manage and improve product quality and yield
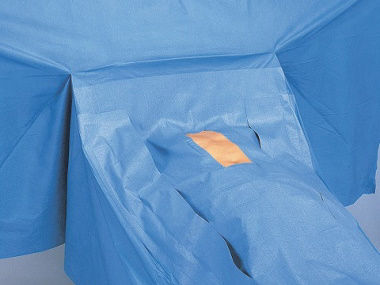
Drape is provided with the sterilization process or non sterilization process. Sterile Drape is designed to provide physical protection for the patient in both incision and non incision for the patient and in particular to support drape from contamination of surgical incision during surgery. Non Sterile Drape is designed to provide physical protection for the pre and post surgical preparation processes and handling requirement.

Surgical Gown is designed to cover the front, back, and arms of surgeons and nurses in the surgery procedures in order to provide a physical barrier between the patient and surgeons or nurses from contamination. It is provided with the sterilization process because the certain part of the gown like the front and the near the hand from the elbow to the wrist area may have direct contact with the patient during the surgical procedure.
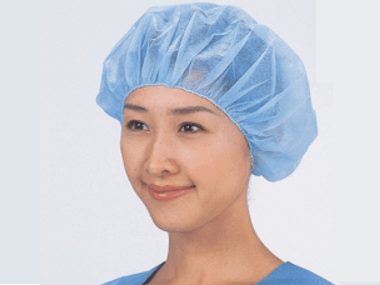
Surgical Cap is provided with the sterilization process or non sterilization process. Surgical Cap is designed to cover hair of the healthcare providers and patients during their patient care activity, to prevent contamination originated from hair or scalp under normal Central Sterile Service Unit (CSSU) surgical procedure. Caps like mask do not require to be steriled before use unless request for certain specific surgery.
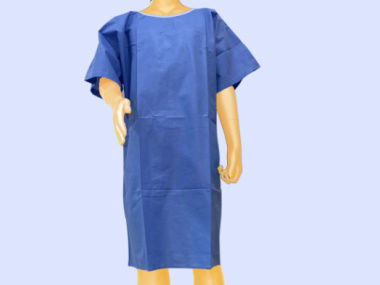
Surgical Supporting Product-SSP is provided with the sterilization process or non sterilization process. Sterile Surgical Supporting Prodcut is designed to support the surgical procedure, which provides the physical protection for the patients, surgeons, nurses and equipments in the surgery from the contamination. Non Sterile Surgical Supporting Product is designed to support process and handling requirement.
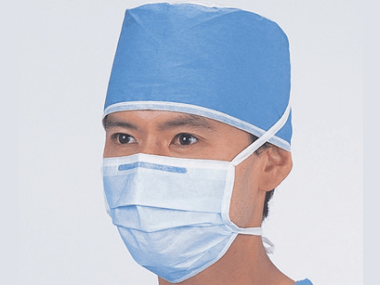
Surgical Mask is designed to be worn by the healthcare providers to protect both patients and healthcare providers against possible microorganisms infection, in blood and body fluids and airborne particulates. Its normally cover the wearer's mouth and nose and eyes for those mask with protective anti-fog transparent seal.

Surgical Pack inclusively content Surgical Drape, Surgical Gown, and Surgical Supporting Product.

Most of our products are designed to be applied in very hygenic environment, such as hospital or operation theater. To be accepted into medical industry means there is very stringent control on the quality of our product.
We had invested heavily to construct a cleanroom production environment, and constantly improve ourselves to meet the industry standard. All our product is guraranteed to adhere with very high industry standard so you can have a peace of mind in using our products.
Please contact our sales people for details specification on each product, customization is possible if there is sufficient production volume in order.
Medipro (Malaysia) Sdn Bhd
Lot 15, Lebuh Hishamuddin 3,
Kaw 20, Bdr Sultan Suleiman,
42000 Port Klang,
Selangor, Malaysia
TEL: 603-3179 0025
FAX: 603-3176 4229
EMAIL: info@medipro.com.my
Medipro (Thailand) Co., Ltd.
55/35 Moo 15 Bangsaothong Sub-District,
Bangsaothong District,
Samut Prakarn Province,
Thailand 10570
TEL: 66 (0)2170-7445-6
FAX: 66 (0)2170-7447